![Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII](https://www.tevagenerics.com/globalassets/products/image//M-N/Meth0727.png)
Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII
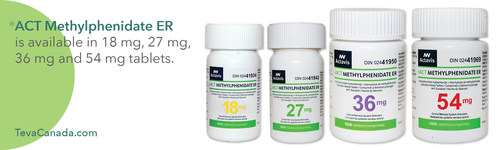
Teva Canada Announces the Launch of <C>ACT Methylphenidate ER® Tablets, a Generic Version of <C>Concerta® for the Treatment of ADHD
![Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII](https://www.tevagenerics.com/globalassets/products/image//M-N/Meth0725.png)
Teva's generic of Concerta® Extended-Release Tablets [CII]: Methylphenidate Hydrochloride Extended-Release Tablets, USP CII

Differences in the In Vitro and In Vivo Pharmacokinetic Profiles of Once-Daily Modified-Release Methylphenidate Formulations in Canada: Examination of Current Bioequivalence Criteria - ScienceDirect