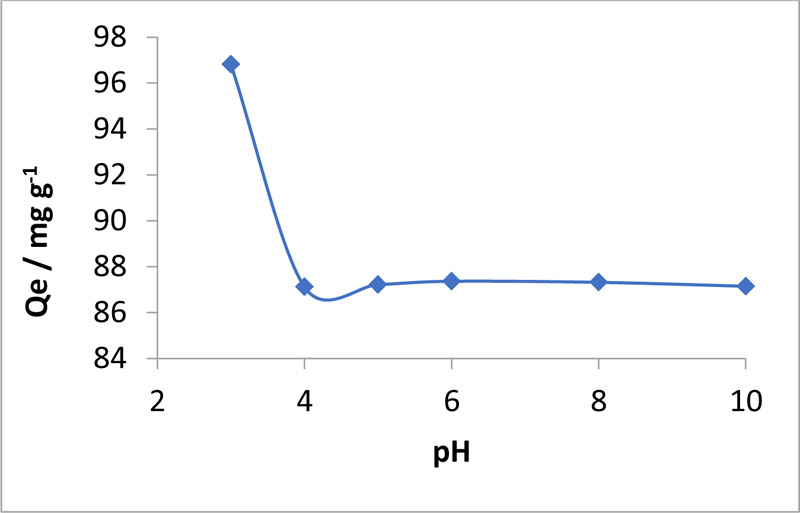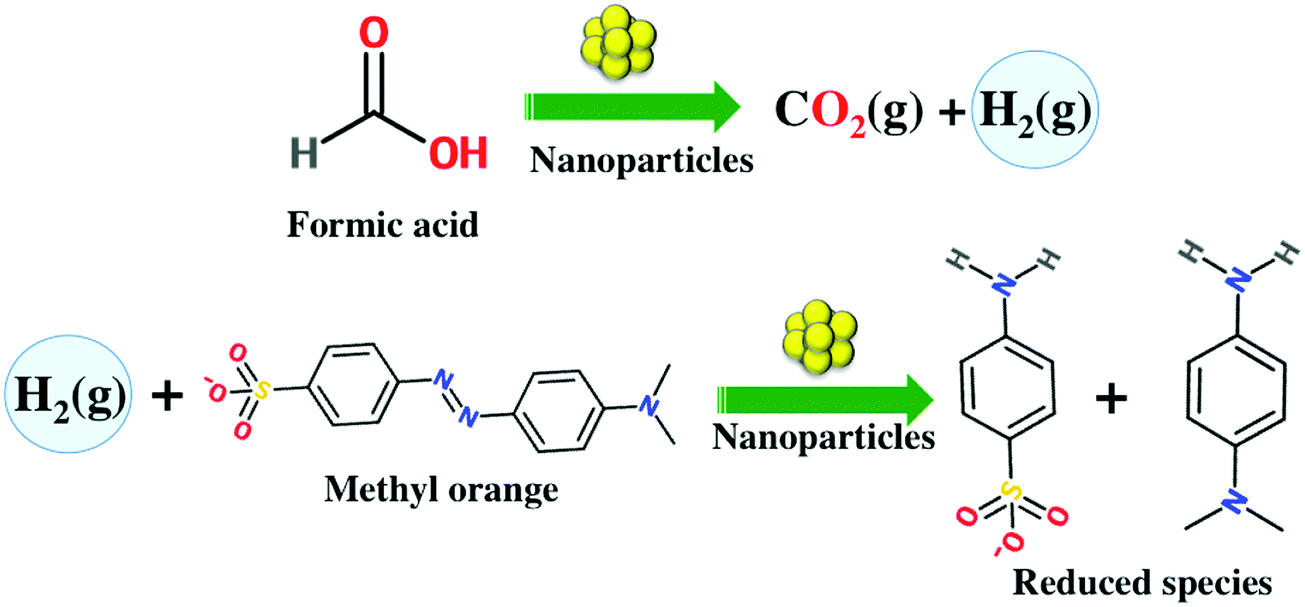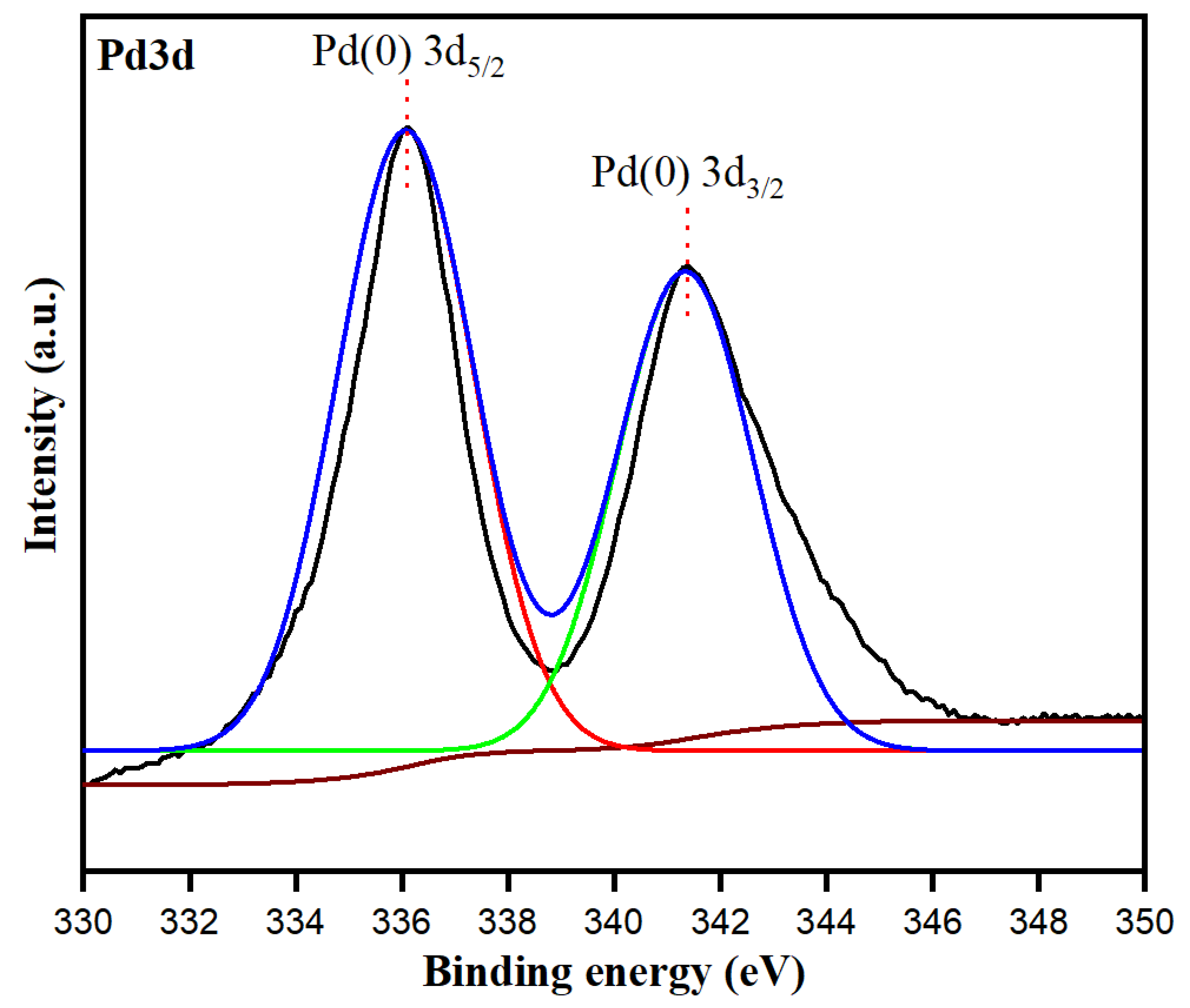
Toxics | Free Full-Text | Bioengineered Matricaria recutita Extract-Assisted Palladium Nanoparticles for the Congo Red Dye Degradation and Catalytic Reduction of 4-Nitrophenol to 4-Aminophenol | HTML

Figure 3 from Rapid degradation of azo dye methyl orange using hollow cobalt nanoparticles. | Semantic Scholar
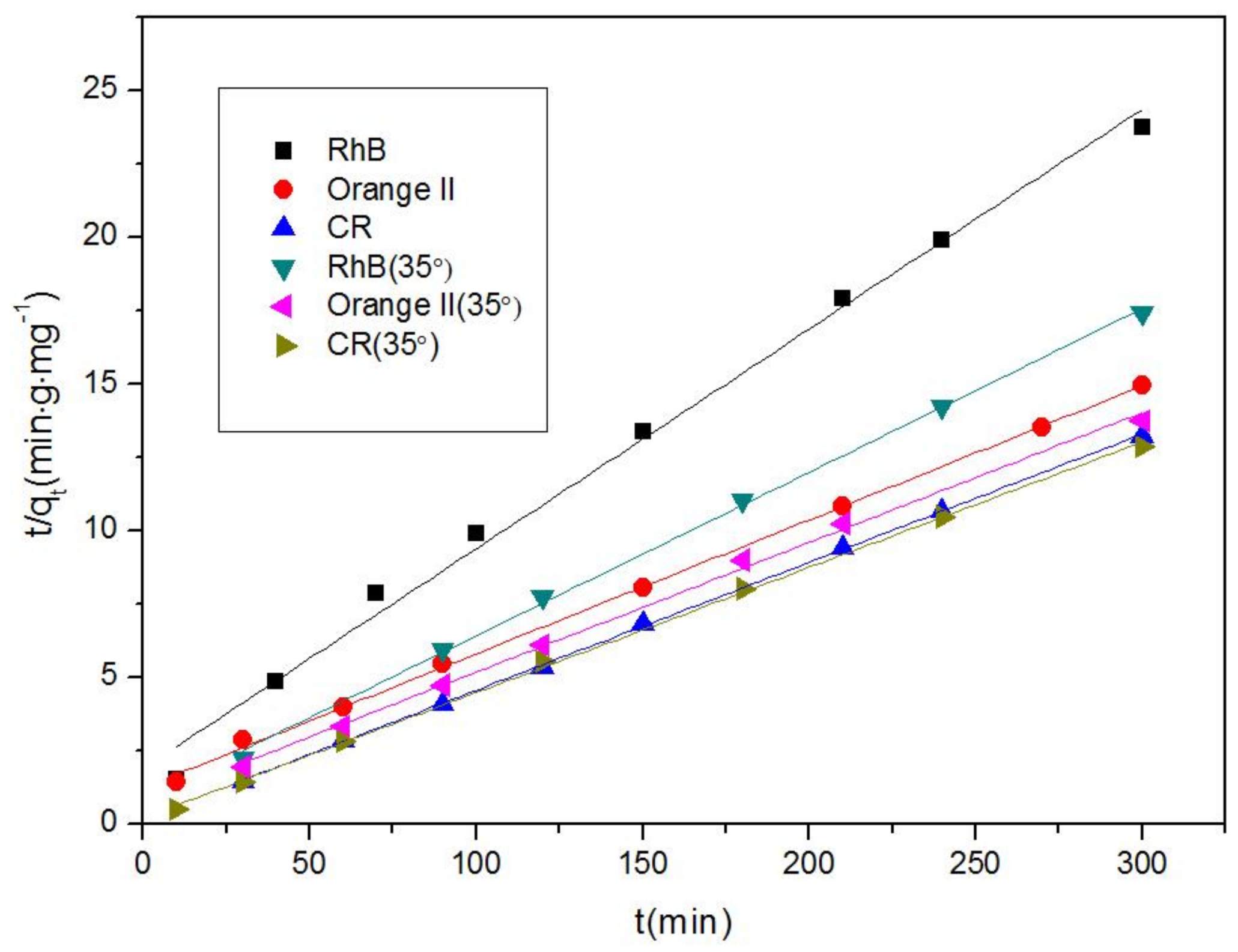
Nanomaterials | Free Full-Text | Comparison Study on the Adsorption Capacity of Rhodamine B, Congo Red, and Orange II on Fe-MOFs | HTML

Efficient degradation of methyl orange in water via both radical and non-radical pathways using Fe-Co bimetal-doped MCM-41 as peroxymonosulfate activator - ScienceDirect

Degradation of Methyl Orange and Congo Red dyes by using TiO2 nanoparticles activated by the solar and the solar-like radiation - ScienceDirect
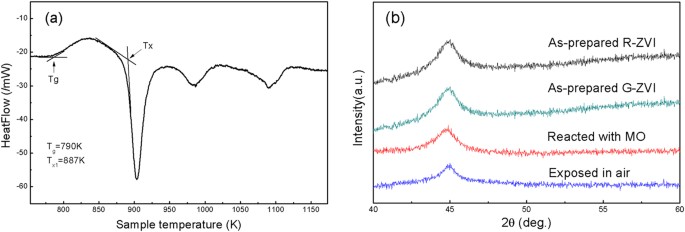
A highly efficient degradation mechanism of methyl orange using Fe-based metallic glass powders | Scientific Reports

Figure 2 from Rapid degradation of azo dye methyl orange using hollow cobalt nanoparticles. | Semantic Scholar
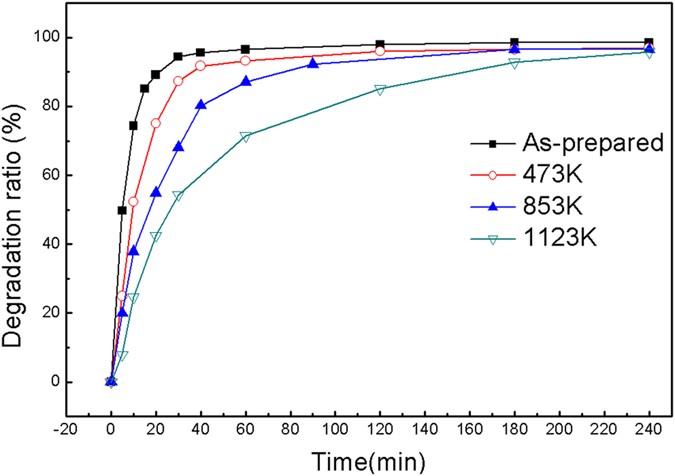
A highly efficient degradation mechanism of methyl orange using Fe-based metallic glass powders | Scientific Reports
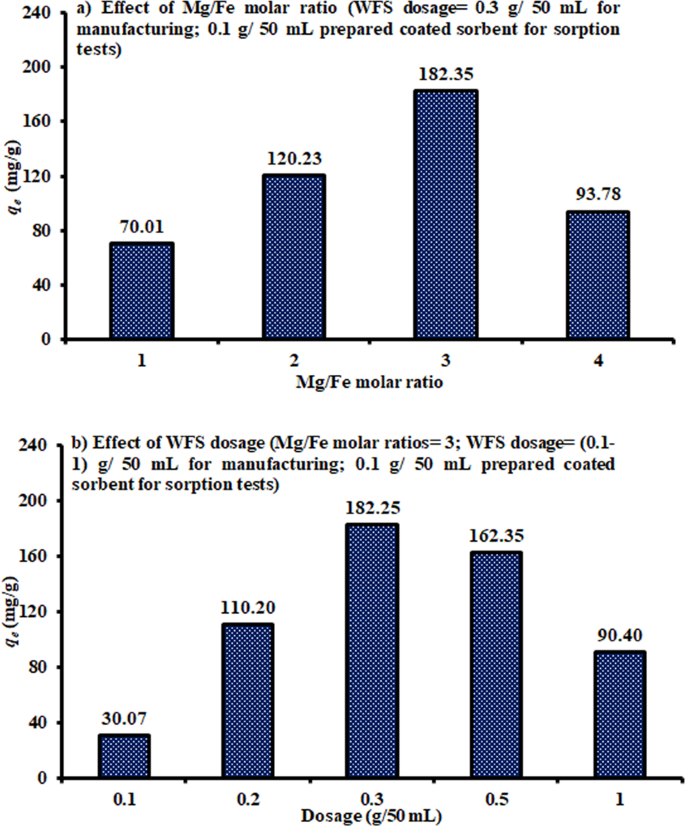
Waste foundry sand/MgFe-layered double hydroxides composite material for efficient removal of Congo red dye from aqueous solution | Scientific Reports

Green synthesis, characterization and catalytic degradation studies of gold nanoparticles against congo red and methyl orange - ScienceDirect

Mechanistic study on methyl orange and congo red adsorption onto polyvinyl pyrrolidone modified magnesium oxide | SpringerLink

UV-Vis absorption spectra for the reduction of Congo Red dye by NaBH 4... | Download Scientific Diagram

Efficient degradation of environmental contaminants using Pd-RGO nanocomposite as a retrievable catalyst | springerprofessional.de

Green synthesis, characterization and catalytic degradation studies of gold nanoparticles against congo red and methyl orange - ScienceDirect

A highly efficient degradation mechanism of methyl orange using Fe-based metallic glass powders – topic of research paper in Nano-technology. Download scholarly article PDF and read for free on CyberLeninka open science

A highly efficient degradation mechanism of methyl orange using Fe-based metallic glass powders | Scientific Reports